S-BOND TECHNOLOGY
How Our Soldering Works
S-Bond® products work with the addition of titanium and/or rare earth elements to conventional solder alloy bases. These active elements migrate to any interface and react or interact with the opposing material surface to either remove oxides and nitrides and transport them into the bulk of the solder as an inert material or adhere to them. This process occurs while S-Bond is molten, after the thin oxide “skin” that forms on the surface of the molten S-Bond is broken, thus allowing contact between the bulk S-Bond and the substrate surface. The breaking of this skin is referred to as “activation” and is done by application of a low level of mechanical shear Sforces at the interface between the S-Bond material and the substrate. The level of shear required is small and can be delivered by brushing or scraping the surface, sliding the joining surfaces relative to one another, or application of high frequency vibration to the parts to be joined. The figures below show two types of mechanical activation, one is brushing or peening and the other is ultrasonic agitation.

Once the surface oxide layer has been disrupted, the S-Bond solder reacts almost instantaneously and in the case of metallurgical bond, irreversibly with the substrate surfaces, to create strong adherence onto the joint surface. The bonded layer at the substrate surface will not be affected, therefore a good interfacial bond strength is maintained and re-activation of the surface is not required.
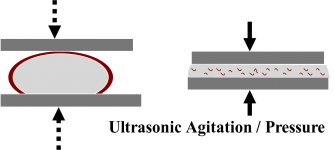
The activation process for S-Bond products is not specifically the mechanism found in conventional ultrasonic soldering, which uses high focused ultrasonic energy directed against the surface of the substrate material to break up the surfaces’ oxide layers and allowing the molten solder to contact clean base material. S-Bond® can adhere to these layers so the mechanical activation is only used to break the oxides on the molten S-Bond surfaces. The oxide layer being broken on the S-Bond® surface is very thin and thus requires very little energy to be disrupted.
A feature of S-Bond products is that they do not flow or wick into openings like conventional solders. Unless pushed, our materials stay where they are placed. This can be useful in situations where precision joining is required.
S-Bond adheres to surfaces utilizing one of two mechanisms shown below.

For S-Bond 220, metallurgical bond mechanism operates with copper and aluminum surfaces when S-Bond joins at 250ºC. The active elements in S-Bond alloys enable the reaction of Sn-Ag-Ti with the underlying base metals to produce a metallurgically reacted joint. This process can also be used on many materials when activated at significantly elevated temperatures, normally as a metallurgical pre-treatment process, to provide a S-Bond metallized surface for solder joining in later operations.
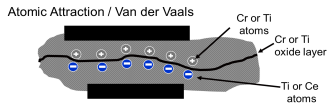
The other adhesive bond mechanism that operates at regular joining temperatures (250°C) is one of adhesive joining or attraction of surfaces with opposite “electronic” charges.


Since metals such as titanium and stainless steel have very thin ‘dielectric’ protective oxides such as TiO2 or Cr3O2, they provide an insulator layer that the S-Bond active elements and the elements in the base metals or ceramics attract across as in typical Van der Waals attractive bonding. The schematic above further illustrates this and how our soldering works.
Contact Us for more information on how our soldering works and refer to our Technology & Applications White Papers section and the Blog.
More Information?
If you would like someone to contact you or provide information, please submit the information below.
