Active solders such as S-Bond have wide application in joining a wide variety of metals including aluminum, copper, stainless steel, titanium, all based on S-Bond alloys’ ability to directly wet and adhere to the metallic surface compounds. Using mechanical activation active solders such as S-Bond successfully join like and dissimilar metal combinations in a wide variety of applications, from heat sinks and sensors to medical/surgical devices.
Soldering of metals has always depended on the “disruption” of surface oxide or other compounds that naturally form on metals. These surface oxides need to be removed for molten solders to permit the liquid solder to “wet” and adhere to metallic surfaces. Thus in soldering, the major process component of soldering is removal of these compounds such that the molten solder can adhere and react with the underlying clean metal surface. In conventional soldering, chemical fluxes are used to remove surface oxides as the solder filler metals melt and flow onto to the surface the flux has just cleaned. Fluxes are nominally acids of differing activity, depending on the metals being soldered and the stability of the oxides that have formed on the metal.
Metals such as copper, nickel and iron naturally form oxides that are not as stable at the oxides that form on aluminum, stainless steel, and titanium and thus the fluxes and/or surface preparations prior to soldering differ. Copper and nickel are easily soldered with milder fluxes and/or rosins. Their oxides are easily reduced. On the other hand, aluminum, stainless steels and titanium, all known for their corrosion resistance (due to their stable surface oxides) are difficult to solder and either employ aggressive acidic fluxes or plating with either nickel or copper prior to soldering. After plating, then these corrosion resistant metals can be soldered with milder fluxes.
S-Bond’s active solders eliminate the flux requirement and the need for plating. With just mechanical activation, S-Bond alloys wet and adhere to all metals. There are two main mechanisms active solders adhere. These mechanisms are illustrated below.
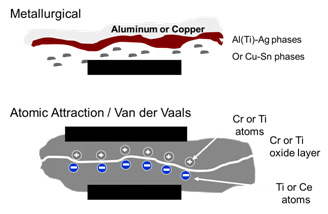
On copper, copper alloys, aluminum and nickel metallurgical bonding occurs as the “active elements (Ti, Ce, Ga and/or Mg) interact with the base metals’ surface oxides, disrupt them and then the S-Bond elements react with the underlying metals to form intermetallic zones that provide the basis for adherence.
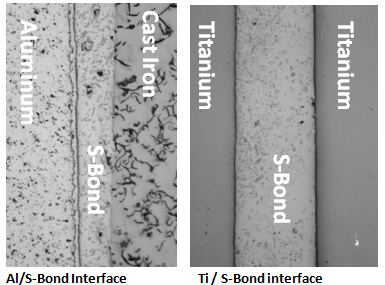
In more corrosion resistant alloys such as Ti and/or stainless steel S-Bond adherence is based in atomic attraction at the very local level. The surface oxide layers are not disrupted, but the “atmospheres” of the active elements in the S-Bond solders interact across the oxide layer and create attractive forces. The images below show the microstructural differences between these two “operative” S-Bond joining mechanisms. The aluminum / S-Bond image illustrates the S-Al-Ag phase that forms at the interface. The Ti / S-Bond interface shows excellent interface conformance but no resolvable reaction zone.
With these two metal bonding mechanisms, S-Bond active solders can join all metals with varying bond strengths. Bond lap shear strengths range from 2,000 – 8,000 psi (14-56 MPa). The bonding is metallic and offers good electrical and thermal conductivity. Another advantage of active soldering is the ability to directly solder dissimilar metals. When conventionally soldering, dissimilar metal joining presents challenges in selecting a compatible flux that can remove oxides on copper and aluminum at the same time while the solder reacts with the surface and many times only plating both sides of the joint is effective for soldering. In S-Bond active soldering the two different adhesion mechanisms operate in parallel on opposite sides of the joint, at the same time without flux, thus S-Bond is a one step process. Additionally, S-Bond joining is done at lower soldering temperatures and thus the process can mitigate the negative effects of thermal expansion mismatch when a soldered assembly cools from the soldering temperatures.
If you would like to evaluate S-Bond for your metal joining applications you may order a Test Kit or Contact Us to discuss the application and/or have us quote making prototype parts.
