Many times our customers have to coat assemblies operations after aluminum bonding, graphite bonding, ceramic to metal bonding, etc; this can present certain challenges that one should be aware of since soldered joints. Unlike welded and many brazed joints, soldered joints utilize a significantly different filler metal. In the case of S-Bond solders, Sn-Ag is the common base filler that is used in aluminum bonding as well as copper, steel, stainless steel, refractory metals, and titanium and many other metals. As such, then the properties of the joint MUST be considered when coating.
The range of coatings seen in industry vary widely, but they can be put in several categories, these are overlay, diffusion or chemical conversion coatings. The overlay coating is the most commonly used in industry. Overlay coating is as it states, it is a coating placed over the base materials and thus over the joint on bonded assemblies (ie. aluminum bonding, graphite bonding, ceramic to metal bonding).
Such overlay coatings include:
- Electroplate
- Powder coat
- Paint
- Thermal Spray
- Vapor deposition (evaporative, sputtering or chemical decomposition)
With the exception of electroplate, S-Bond solder joints are compatible with these processes on the provision the part temperature in the coating process does not exceed the remelt temperature of the S-Bond (solder) joint, in the range of 180 – 450C, depending in the solder alloy used. Electroplate coatings can be made over solder joints as long as the preparation of surfaces for electroplate ( many times acid etching is used) does not preferentially attack the solders significantly faster that the surrounding base materials, recognizing that thin joints of less “noble” metals (Sn, Zn, etc.) adjacent more noble metals like aluminum, stainless steel, nickel alloys (platinum being one of the most noble) can set up an galvanic corrosion cell as illustrated in Figure 1.
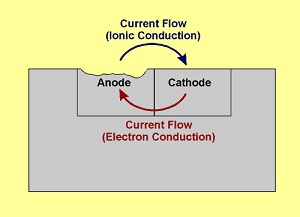
This situation can arise in service, in a plating or in an acid cleaning bath and can be managed paying attention to the time or acid activity in order to prevent excessive chemical attack (corrosion reaction) with the solder interface. S-Bond joints between aluminum, copper and a wide range of metals has been successfully electroplated, once the plater pays attention to the dissimilar metal combination a soldered assembly presents.
The other set of coating technologies that can be applied to bonded assemblies are the conversion coatings… of which two common industrial coatings for aluminum include chromate coating or anodizing. Anodizing is an electrolytic passivation process used to increase the thickness of the natural oxide layer on the surface of aluminum parts. The process is called “anodizing” because the part to be treated forms the anode electrode of an electrical circuit. Anodizing increases corrosion resistance and wear resistance. Anodic films are most commonly applied to protect aluminum alloys, although processes also exist for titanium, zinc, magnesium, niobium, and tantalum. Chromate conversion coatings are a type of conversion coating used to passivate aluminum, zinc, cadmium, copper, silver, magnesium, and tin alloys and is primarily used as a corrosion inhibitor. The process is named after the chromate found in the chromic acid used in the bath, more commonly known as hexavalent chromium.
NOTE: S-Bond bonded assemblies CANNOT be anodized since this chemical conversion process includes the use of very aggressive acids and depends on the base aluminum to chemically convert to Al2O3 (aluminum oxide) … in the area of joint where S-Bond would be exposed to the chemical conversion acidic bath, the Sn-Ag would be aggressively attached and furthermore the lack of aluminum locally would not enable to support the conversion to the anodized Al2O3 coating, thus providing a local spot for corrosion.
On the other hand S-Bond joined parts are compatible with chromate conversion coating as long as the pre-cleaning steps to not expose the S-Bond solder interface to a long duration acid dip where the Sn-Ag interface is attached preferentially. The chromate coating will take to the S-Bond layer and provide corrosion protection for the S-Bond filler metal, thus protecting the whole assembly.
Diffusion coatings such as carburizing, nitriding or aluminizing are thermally activated coating processes where higher concentrations of the coating elements (C, N2 or Al) are diffused from vapor, liquids or solids rich in those elements into the base metals. These processes generally occur above 800°C, well above the S-Bond remelt temperatures so they would not be compatible with S-Bond joined assemblies.
In summary; S-Bond is compatible with many overlay and some selected conversion coatings. We recommend the following post bond assembly coating processes… chose the ones that provide the type of protection required:
- Powder coatings
- Painting
- Ni, Au, Sn, Zn Electroplates (provided acid cleaning is controlled)
- Chromate conversion coatings
- Vapor deposited coatings (provided assembly remains below (250°C)
Please Contact Us, if there are questions on coating your bonded assemblies.
